
Back pressure can be created naturally as on the right side of Figure 4. Reverse osmosis and reverse dialysis(also called filtration) are processes that occur when back pressure is sufficient to reverse the normal direction of substances through membranes. (b) The fluid level rises until the back pressure ρgh equals the relative osmotic pressure then, the net transfer of water is zero. 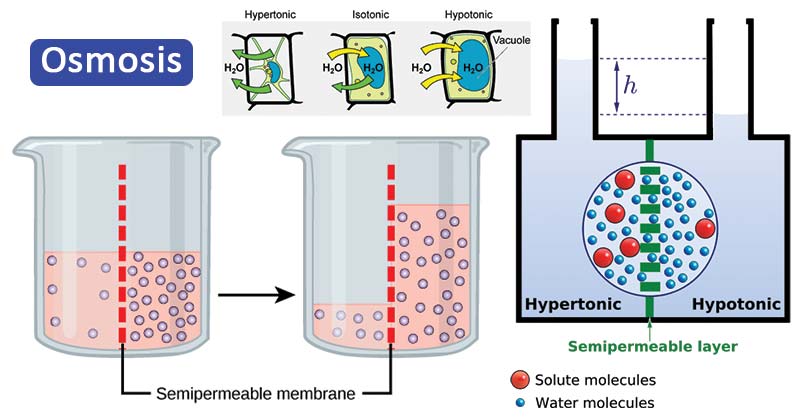
After they were put it either sugar water or plain water. Two of the cells were filled with concentrated sugar water and one was filled with water. 
In this lab three model cells were made from dialysis tubing. Osmosis will be to the right, since water is less concentrated there. The effects of osmosis on cells that are put in solutions that have different concentration Abstract: In this lab experiment three model cells were placed in different solution. (a) Two sugar-water solutions of different concentrations, separated by a semipermeable membrane that passes water but not sugar. Dialysis can similarly cause substantial pressures. Turgor describes the condition of a plant in which the fluid in a cell exerts a pressure against the cell wall. This value means that water will diffuse through the membrane until the salt water surface rises 268 m above the pure-water surface! One example of pressure created by osmosis is turgor in plants (many wilt when too dry). For example, if pure water and sea water are separated by a semipermeable membrane that passes no salt, osmotic pressure will be 25.9 atm. Osmotic pressure can be large, depending on the size of the concentration difference. It can be shown that the average distance\boldsymbolthat stops osmosis is also called the relative osmotic pressure if neither solution is pure water, and it is called the osmotic pressure if one solution is pure water. The densities of common materials are great enough that molecules cannot travel very far before having a collision that can scatter them in any direction, including straight backward. Fluids, like fish fumes or odors entering ice cubes, can even diffuse through solids.ĭiffusion is a slow process over macroscopic distances. Diffusion is the movement of substances due to random thermal molecular motion. This motion is called a random walk and is illustrated in Figure 1. In fluids they move about randomly even in the absence of macroscopic flow. Atoms and molecules are in constant motion at any temperature. There is something fishy about the ice cube from your freezer-how did it pick up those food odors? How does soaking a sprained ankle in Epsom salt reduce swelling? The answer to these questions are related to atomic and molecular transport phenomena-another mode of fluid motion. Define diffusion, osmosis, dialysis, and active transport.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
